
What is it – Transesterification
Do you know how biodiesel, a renewable and environmentally friendly alternative to petroleum diesel, is made? Today, FUELSNews will be covering transesterification, the process of creating biodiesel. We’ll also discuss how long the process takes and how it differs from traditional petroleum diesel refining. So, let’s dive into the fascinating world of transesterification and discover the potential of this sustainable fuel source.
What is Transesterification?
Transesterification is a chemical process used to produce biodiesel from renewable sources like vegetable oils, animal fats, or waste cooking oils. It involves the conversion of these lipid feedstocks into a more suitable form for use as a fuel in diesel engines.
The process works by reacting the feedstock with an alcohol, typically methanol or ethanol, in the presence of a catalyst, usually an alkali like sodium hydroxide (NaOH) or potassium hydroxide (KOH). The reaction breaks the ester bonds in the triglycerides present in the feedstock and replaces them with alcohol molecules. This results in the formation of mono-alkyl esters, which are the main components of biodiesel, and glycerol as a byproduct.
The overall transesterification reaction can be summarized as follows:
Triglyceride + Alcohol → Mono-alkyl esters (biodiesel) + Glycerol
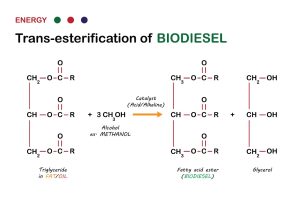
Biodiesel produced through transesterification has several advantages over traditional petroleum diesel, including lower emissions, improved lubricity, and biodegradability. Additionally, biodiesel can be blended with petroleum diesel in varying proportions, allowing for a smoother transition to renewable fuels and reducing dependency on fossil fuels. It’s worth noting that biodiesel is not a fuel itself, but technically a mono-alkyl ester that can be blended with fuel. For that reason, you cannot use 100% biodiesel in your engine. Still, most engine manufacturers approve up to a B20 use in engines.
How long does transesterification take?
A typical transesterification process can take anywhere from a few minutes to several hours, depending on the type of feedstock, the alcohol and catalyst used, the reaction temperature, and the agitation or mixing technique employed. In most small-scale and commercial biodiesel production systems, transesterification is typically carried out at temperatures ranging from 50 to 70 degrees Celsius (122 to 158 degrees Fahrenheit). Under these conditions, the reaction time can range from 30 minutes to 2 hours to achieve sufficient conversion of the feedstock into biodiesel.
After the transesterification reaction is complete, additional time is needed for the separation of the biodiesel, glycerol, and any unreacted chemicals or impurities. This separation step can take several hours to days, depending on the method used, such as gravity settling, centrifugation, or membrane filtration.
What is the difference between transesterification and a normal petroleum diesel refining process?
Petroleum diesel refining and biodiesel transesterification are two distinct processes used to produce different types of fuels. The main differences between the two processes can be categorized in terms of feedstock origin, production process, environmental impact, energy content, and compatibility.
The feedstock origin for petroleum diesel is crude oil, which is a fossil fuel. In contrast, biodiesel is produced from renewable sources, which can be replenished over a much shorter timeframe.
Petroleum diesel refining involves the separation and processing of various hydrocarbon fractions from crude oil. This process typically includes steps like desalting, distillation, cracking, and hydrotreating. Biodiesel transesterification, on the other hand, is a chemical reaction that converts lipid feedstocks into mono-alkyl esters (biodiesel) and glycerol.
If you are looking into more sustainable fuel options for your business, Mansfield has you covered. Visit our website to learn more about renewable fuel options and how we can help you on the road to net zero.
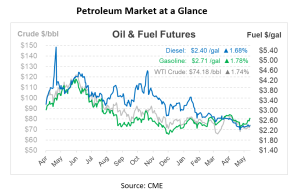
This article is part of Daily Market News & Insights
Tagged: Biodiesel, biofuels, sustainability, Transesterification
MARKET CONDITION REPORT - DISCLAIMER
The information contained herein is derived from sources believed to be reliable; however, this information is not guaranteed as to its accuracy or completeness. Furthermore, no responsibility is assumed for use of this material and no express or implied warranties or guarantees are made. This material and any view or comment expressed herein are provided for informational purposes only and should not be construed in any way as an inducement or recommendation to buy or sell products, commodity futures or options contracts.





